Onyx Vantage 9590 Specifications
Oxygen Saturation Display Range: 0% to 100% SpO2
Pulse Rate Display Range: 18 to 321 beats per minute (BPM)
Declared Accuracy:
The tables below show Arms values measured using the Onyx Vantage 9590 in a clinical study in non-motion conditions.
Accuracy Summary by Decade – Finger and Thumb
| Decade | Oxygen Saturation (Arms) |
|---|---|
| 70 – 80% | ±2 |
| 80 – 90% | ±2 |
| 90 – 100% | ±2 |
| 70 – 100% | ±2 |
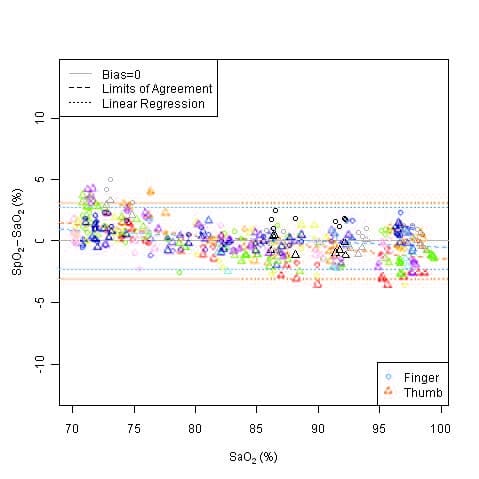
This graph shows plots of the error (SpO2 – SaO2) by SaO2 using the 9590 with a linear regression fit and upper 95% and lower 95% limits of agreement. Each sample data point is identified by subject from a clinical study in non-motion conditions.
Accuracy Summary by Decade – Toe
| Decade | Oxygen Saturation (Arms) |
|---|---|
| 70 – 80% | ±2 |
| 80 – 90% | ±3 |
| 90 – 100% | ±3 |
| 70 – 100% | ±3 |
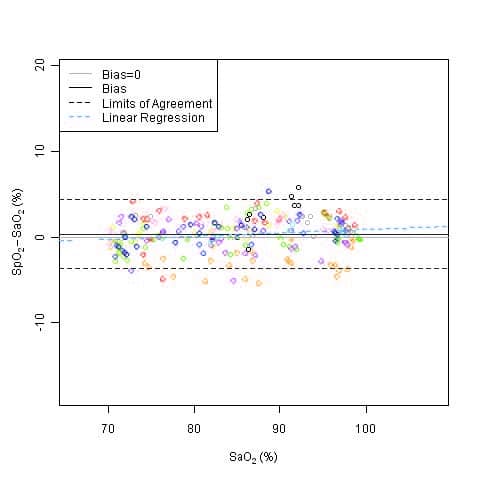
This graph shows plots of the error (SpO2 – SaO2) by SaO2 using the 9590 with a linear regression fit and upper 95% and lower 95% limits of agreement. Each sample data point is identified by subject from a clinical study using toes in non-motion conditions.
SpO2 Low Perfusion Accuracy (Arms*) 70 to 100% ±2 digits
Pulse Rate Declared Accuracy Range (Arms*): 20 to 250 BPM ±3 digits
Low Perfusion Pulse Rate Declared Accuracy Range (Arms*): 40 to 240 BPM ±3 digits
Measurement Wavelengths and Output Power**:
Red: 660 nanometers @ 0.8 mW maximum average
Infrared: 910 nanometers @ 1.2 mW maximum average
Temperature:
Operating: 23 °F to 104 °F / -5 °C to 40 °C
Storage/Transportation: -40 °F to 158 °F / -40 °C to 70 °C
Time (from storage) for monitor to be ready for its intended use: 3 minutes to warm from -40 °C to -5 °C
5 minutes to cool from 70 °C to 40 °C
Humidity:
Operating: 10% to 90% non-condensing
Storage/Transportation: 10% to 95% non-condensing
Altitude:
Operating: Up to 13,123 feet / 4,000 meters
Hyperbaric Pressure: Up to 4 atmospheres
Battery Life:
Operating: Approximately 6,000 spot checks or 36 hours of continuous operation
using new alkaline batteries.
Storage: 12 months
Classifications per ANSI/AAMI ES60601-1 / CAN/CSA-C22.2 No. 60601-1:
Degree of Protection: Type BF-Applied Part
Enclosure Degree of Ingress Protection: IP32
Mode of Operation: Continuous
This product complies with ISO 10993-1, Biological evaluation of medical devices – Part 1: Evaluation and testing.
This device is not made with natural rubber latex.
*± 1 Arms represents approximately 68% of measurements at zero bias.
**This information is especially useful for clinicians performing photodynamic therapy.
8339-001-05

