Resources
-
Understanding Accuracy, Bias, and Variability in SpO2 Readings View
-
Accuracy and Superior Performance of PureSAT and PureLight Oximetry Technologies View
-
Fingertip Pulse Oximeter Performance in Dyspnea and Low Perfusion During Hypoxic Events View
-
Accurate Oximetry Monitoring – See the Technology Difference View
Onyx3 9591 Specifications
Oxygen Saturation Display Range: 0% to 100% SpO2
Pulse Rate Display Range: 18 to 321 beats per minute (BPM)
Respiration Rate Display Range: 2 to 80 breaths per minute
Declared Accuracy*: The table below shows Arms values measured using the Model 9591 in non-motion conditions.
| Range | Specified Oxygen Saturation (Arms) | Finger Oxygen Saturation (Arms) | 95% Limits of Agreement | Low Perfusion Oxygen Saturation (Arms) |
|---|---|---|---|---|
| 70 – 100% | ±2 | ± 1.31 | (-2.3 – 2.9) | ±2 |
| 70 – 80% | ±2 | ± 1.62 | (-1.7 – 3.7) | ±2 |
| 80 – 90% | ±2 | ± 1.05 | (-2.3 – 2.2) | ±2 |
| 90 – 100% | ±2 | ± 1.18 | (-2.4 – 2.4) | ±2 |
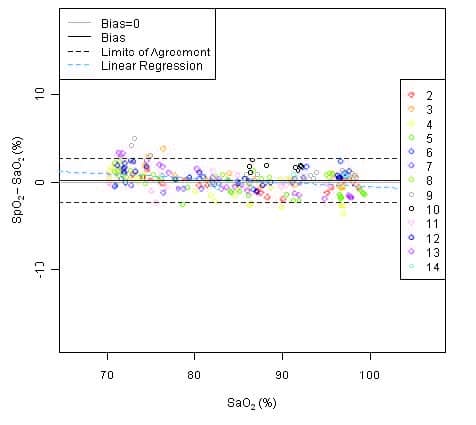
This graph shows plots of the error (SpO2 – SaO2) by SaO2 using the Model 9591 with a linear regression fit and upper 95% and lower 95% limits of agreement. Each sample data point is identified by subject from a clinical study in non-motion.
SpO2 Low Perfusion Accuracy (Arms)*: 70 to 100% ±2 digits
Pulse Rate Declared Accuracy Range (Arms)*: 20 to 250 BPM ±3 digits
Low Perfusion Pulse Rate Declared Accuracy Range (Arms)*: 40 to 240 BPM ±3 digits
Respiration Rate Accuracy: (Mean Error): 3-44 ± 1 Breath per minute
Measurement Wavelengths and Output Power**:
Red: 660 nanometers @ 0.8 mW max. average
Infrared: 910 nanometers @ 1.2 mW max. average
Temperature:
Operating: -5 °C to 40 °C / 23 °F to 104 °F
Storage/Transportation: -40 °C to 70 °C / -40 °F to 158 °F
Time (from storage) for monitor to be ready for its intended use: 3 minutes to warm from -40 °C to -5 °C
5 minutes to cool from 70 °C to 40 °C
Humidity:
Operating: 10% to 95% non-condensing
Storage/Transportation: 10% to 95% non-condensing
Altitude:
Operating: Up to 4,000 meters / 13,123 feet
Hyperbaric Pressure: Up to 4 atmospheres
Battery Life:
Operating: Approximately 2,000 spot checks, or 25 hours of continuous operation using new alkaline batteries.
Storage: 1 month, with batteries installed. CAUTION: Remove batteries if the
device will be stored for more than 30 days.
Classifications per IEC 60601-1/UL 60601-1 / CAN/CSA-C22.2 No. 601.1:
Degree of Protection: Type BF-Applied Part
Enclosure Degree of Ingress Protection: IP32
Mode of Operation: Continuous
This product complies with ISO 10993-1, Biological evaluation of medical devices – Part 1:
Evaluation and testing.
This device is not made with natural rubber latex.
This product complies with RoHS.
This product complies with REACH.
* ±1 Arms represents approximately 68% of measurements.
** This information is especially useful for clinicians performing photodynamic therapy.
Nonin
Locations
Nonin Medical Inc
(Global Headquarters)
Address
13700 1st Ave N,
Plymouth, MN 55441
Email
info@nonin.com
Toll Free (USA and Canada)
1.800.356.8874
Phone
1.763.553.9968
Nonin Medical B.V.
(Europe)
Address
Doctor Paul Janssenweg 150
5026 RH Tilburg
Netherlands
Email
info@nonin.com
